Getting a COVID vaccine just got more complicated after new FDA restrictions
The FDA narrowed approval of the new COVID-19 vaccines.
The U.S. Food and Drug Administration in August narrowed approval of the new, updated COVID-19 vaccines only to people who are over 65, or younger people with underlying conditions that put them at higher risk for severe disease.
That decision has raised questions about access for the millions of Americans who don't fall in those categories -- and potentially even for some who do.
According to pharmacists, insurance groups and trade organizations, it will be more difficult for younger, otherwise healthy people to get a COVID-19 vaccine ahead of the winter respiratory virus season, as it will involve a trip to the doctor instead of walking into a pharmacy, and insurance coverage is, for now, unclear.
And for at least the next few weeks, as the policy shifts are absorbed, the confusion may also impact access for older, higher-risk people, who some pharmacies are still requiring to get a prescription in order to get their vaccines.
This comes as Trump administration officials maintain that "100% of adults in this country can still get the vaccine if they choose," as FDA Commissioner Marty Makary wrote on X. "We are not limiting availability to anyone," he said.
"The FDA's decision does not affect the availability of COVID vaccines for Americans who want them," White House press secretary Karoline Leavitt said Thursday during a press conference. "We believe in individual choice. That's a promise both the president and [HHS Secretary Robert F. Kennedy Jr.] have made. And it's a promise they have now delivered on."
Experts interviewed by ABC News said this may be technically accurate, as a doctor can still determine that a non-eligible person needs the vaccine "off label" and can give them the shot.
But in practice, the confusion around the new policy and fear from doctors of going against the federal guidelines could make it significantly more challenging for people to get that prescription.

It also creates another hurdle to getting the vaccine, if someone needs to add in the step of going to their doctor rather than a pharmacy. The vast majority of Americans have gotten their COVID vaccines from pharmacies, not the doctor's office.
"It's a mess," said Dorit Reiss, a professor at University of California College of the Law, San Francisco, who focuses on vaccine law.
"The starting point is it's a mess. And by the way, that's going to probably decrease uptake all by itself, because people aren't going to know," she said.
For years, health officials have encouraged virtually all Americans to get annual COVID shots, akin to the annual flu vaccine. Under President Donald Trump, health officials have made the case that the benefits of COVID vaccination are clearer for older, high-risk Americans, and there needs to be more research on the benefits for young and healthy people.
COVID vaccine uptake has been low, with just 23 percent of adults and 13 percent of children reported getting an updated COVID vaccine last season, according to the Centers for Disease Control and Prevention.
About 49% of adults 75 years and older had received the latest COVID-19 vaccine as of December 2024, the CDC reported last year.
The laws are different in each state. Where can you get a COVID vaccine where you live?
There are certain states where pharmacists are permitted to only give vaccines to people who qualify under FDA and CDC guidelines, according to Brigid Groves, vice president of professional affairs at the American Pharmacist Association.
That means that it's less likely pharmacies in those states will still offer the shot to people who don't qualify under the new FDA approvals, because pharmacists could open themselves up to liability if they give the vaccine "off label," said Groves.
A handful of states like Massachusetts, New Jersey, Pennsylvania and New Mexico have recently changed their laws to allow pharmacists to give the COVID vaccine despite the FDA change, hoping to keep the status quo in their states -- but the patchwork of changing laws around the country has been difficult to track.
"We're concerned that that pharmacy is no longer going to be able to service the patient population because of the limits that have been placed and the narrowing indication that has occurred," said Groves.
This reality is playing out at CVS pharmacies nationwide, which is the largest pharmacy chain in the country.
In 41 states, CVS will continue to offer COVID vaccines for those who fall under the new FDA authorizations.
But in nine states plus D.C., people will need a prescription to get a COVID shot, even if they meet those qualifications. Those states are: Arizona, Florida, Georgia, Louisiana, Maine, North Carolina, Utah, Oregon and West Virginia, and the District of Columbia.
Walgreens, another large pharmacy chain, said it would give shots where it was legally able to do so, but didn't specify which states would have which rules.
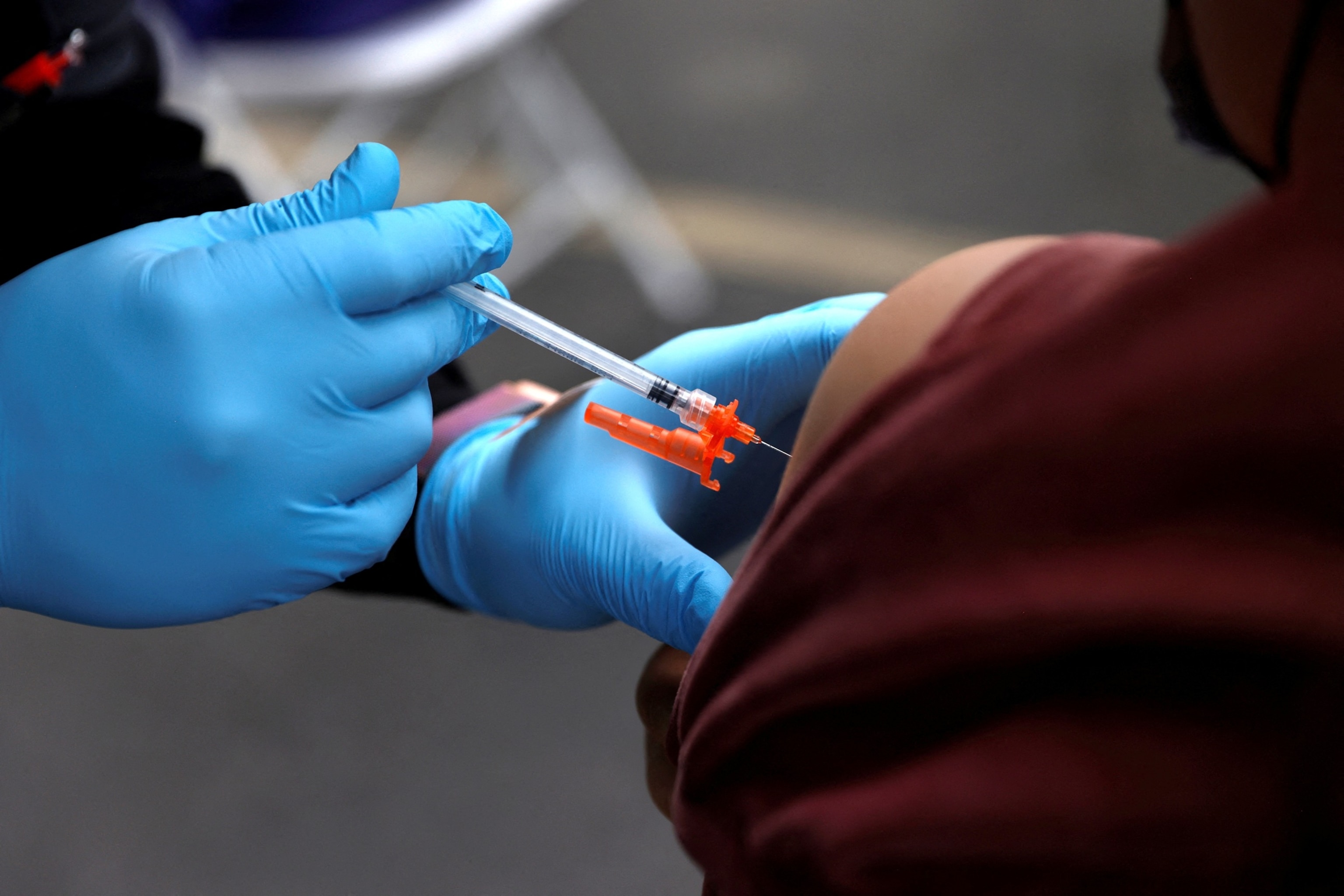
If you're young and healthy, and you want a COVID vaccine, will it be covered?
When it comes to insurance coverage, private insurance providers have suggested they could continue to cover the cost of COVID vaccines for anyone who gets a shot, even for younger, otherwise healthy adults -- although they are awaiting guidance from the CDC's advisory panel, which meets Sept. 18 and 19.
That leaves a few weeks of limbo, just before the fall and winter when cases of COVID could rise.
The meeting is also potentially up in the air. After high-profile departures from officials at the CDC and a public fallout between Kennedy and the since-fired CDC director, Susan Monarez, over changing COVID vaccine policy, Republican Sen. Bill Cassidy, chair of the Senate committee on health, called for it to be postponed.
Insurance providers are watching and waiting, said Tina Stow, spokesperson for America's Health Insurance Plans, the trade group for the health insurance industry.
"Individual health plans and plan sponsors will be prepared to make coverage decisions informed by science, the latest medical evidence and data," Stow said.
Insurers don't have to only cover vaccines recommended by the CDC, but they're still planning to wait and see just what the recommendations are — particularly because the committee, which is made up of all new members handpicked by Kennedy, is less predictable than in the past.
"They might decide that it's cost effective for them to do it," Reiss said. "They may, but they might not. And we may see variation [by provider]."
It's not clear what would happen for people covered by government-funded insurance like Medicare and Medicaid -- which is tied to the CDC recommendations, or the federal program that funds vaccines for low income children, Vaccines For Children.
There is also the question of access for healthy children under 5, who are no longer approved for any COVID vaccines under the FDA's latest decision. Previously, they were authorized for the Pfizer vaccine, which was available to kids as young as 6 months.



